News Highlights
Upcoming Events
Room C13 (IST); Room F405 (FEUP) |
In our workshop on "How to Prepare and Write a Scientific Paper," we embark on a journey int
Room C13 (IST); Room F405 (FEUP) |
In an era where the ability to captivate an audience is a valuable skill, our "How to Prepar
Hybrid: LTI of the Mining Department and Zoom |
University of Azores, Ponta Delgada, Portugal |
Research Groups
The Energy Group develops research focused on hydrocarbons, both from conventional and unconventional sources, low-carbon energy, biofuels and energy efficiency.
The Environment Group consists of a multidisciplinary team of academics, research staff and postgraduates, internationally recognized through the&nb
The Raw Materials Group brings together researchers from different fields of expertise focusing on three main areas: primary and secondary mineral raw materials; primary and secondary n
Featured Projects
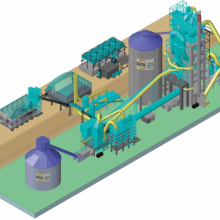
The Clean Cement Line aims to develop and demonstrate a new cement production technology.
The overall goal of IProPBio is to exchange complementary theoretical and experimental knowledge
The Serra da Estrela Nat
It is estimated that 80% of all floating debris are marine plastics.